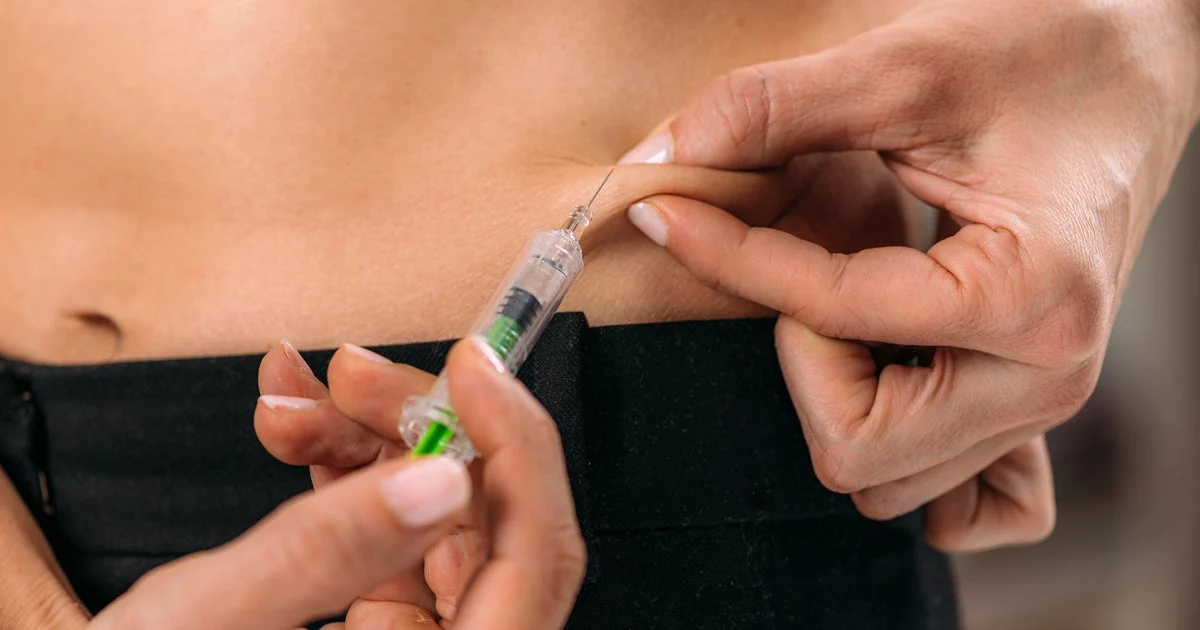
Peptides, short chains of amino acids naturally found in the human body, have surged in popularity online with claims that they can heal injuries, improve anti-aging, and boost overall health. However, medical experts caution that many of these peptides are unapproved by the Food and Drug Administration (FDA) and lack reputable clinical evidence supporting their safety and effectiveness.
While peptides play crucial roles in cellular functions and are ingredients in approved treatments such as insulin and medications like Ozempic and Wegovy, the peptides promoted in viral videos and social media posts often extend well beyond these established uses. Claims circulating on platforms like TikTok and Instagram promise benefits ranging from DNA repair to stress reduction, frequently supported by anecdotal success videos labeled as “peptide transformations.”
Dr. Jon LaPook, CBS News chief medical correspondent, explains that although animal studies in rats and mice suggest some peptides might affect cell function, there are no reproducible, randomized human trials confirming these effects. Many peptides sold online come from unregulated sources and are marketed under the guise of “research use only,” with uncertain quality and safety. Consumers often order these compounds on websites that lack transparency or regulatory oversight and then self-administer them via injection.
Harvard Public Health expert Dr. Monica Wang describes the broad marketing of peptides as a “red flag,” underscoring that products not subjected to rigorous FDA approval or clinical trials frequently make unsupported health claims. She attributes the peptides’ popularity to people’s desire for simple, fast solutions to complex health issues, amplified by social media algorithms and e-commerce platforms presenting such products as easy fixes.
The surge in peptide interest is reflected in search and social media activity: U.S. Google searches related to peptides reached over 10 million in January 2026, growing especially fast in categories linked to anti-aging and metabolic health. The hashtag #peptides on TikTok and Instagram is featured in hundreds of thousands of posts, some amassing millions of views.
Prominent voices such as Health and Human Services Secretary Robert F. Kennedy Jr. have publicly supported peptide use. On a recent podcast, he described personal benefits from peptide treatment and expressed intent to increase access to certain peptides currently restricted by compounding pharmacy regulations. This has prompted questions from experts about what safety and efficacy evaluations would accompany broader availability under FDA oversight.
Why it matters
The rise of unregulated peptide use poses potential safety risks due to the absence of standardized manufacturing, dosing controls, and clinical validation. Consumers self-administering these substances without medical supervision may face unknown health hazards. The growing demand also pressures regulatory agencies to consider how to balance access with protections, highlighting the need for rigorous research and public education.
Background
Peptides are recognized biochemical compounds composed of amino acids linked by chemical bonds. Endogenously produced peptides regulate numerous physiological processes, and several peptide-based therapies have FDA approval for conditions like diabetes and obesity. However, the “wild west” market for peptides—especially those labeled “for research use”—operates largely outside regulatory frameworks, leaving safety and efficacy claims largely unverified. The increased visibility of peptides in consumer health conversations underscores broader trends in direct-to-consumer biohacking and alternative wellness sectors.
Read more Climate & Environment stories on Goka World News.