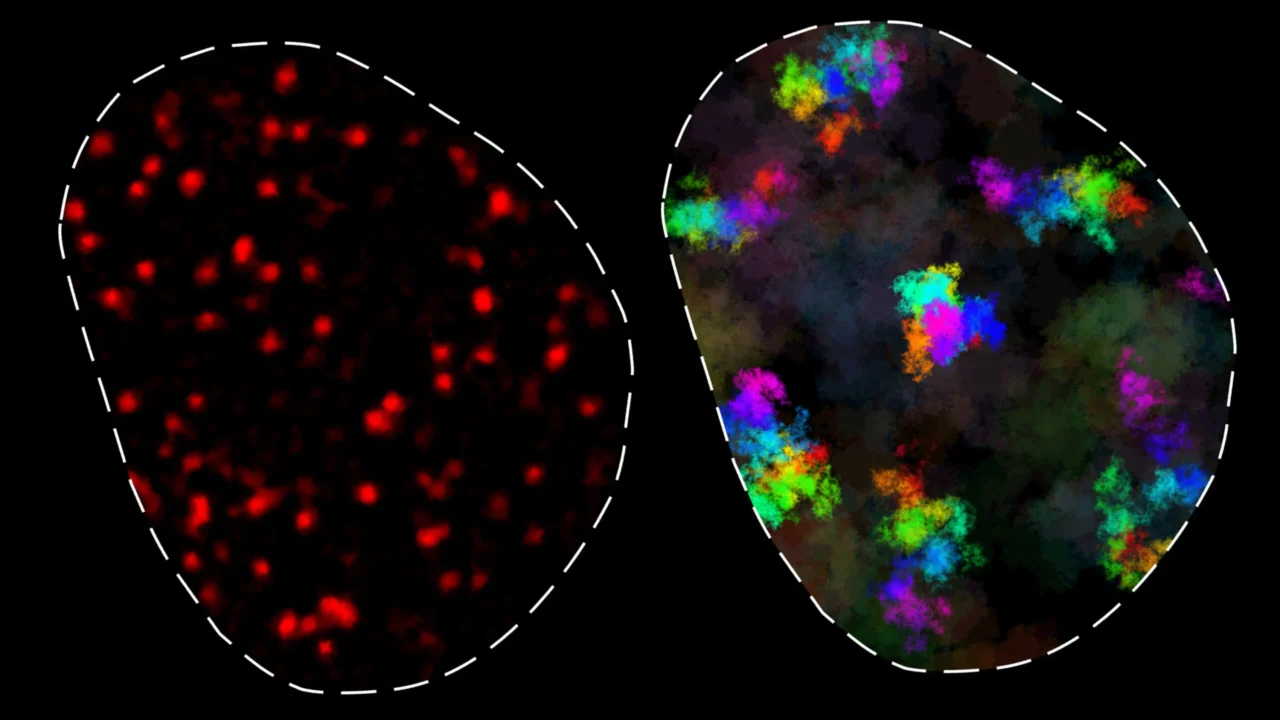
MIT researchers have quantified chromatin movement within living cells at unprecedented timescales, revealing two distinct types of chromatin dynamics that help regulate gene expression and DNA repair. The study, published in Nature Structural and Molecular Biology, used super-resolution microscopy to track chromatin behavior from hundreds of microseconds to several hours.
Mapping chromatin motion across time
Chromatin, a complex of DNA and proteins inside the cell nucleus, constantly moves to enable contact between genes and distant regulatory elements such as enhancers. These movements are critical for gene regulation and facilitating DNA repair by bringing broken DNA ends together. However, chromatin’s behavior is constrained because DNA is a polymer, which pulls parts back after movement.
Previous attempts to measure chromatin movement lacked sufficient temporal range or resolution, leading to inconsistent findings. The MIT team applied MINFLUX, a cutting-edge super-resolution light microscopy technique developed by Nobel laureate Stefan Hell, to overcome these limitations. This enabled tracking of chromatin loci over four orders of magnitude in time (from 200 microseconds to 10 seconds), which, combined with other imaging methods, extended the dynamic range to cover timescales from microseconds to hours.
Constrained versus freer chromatin movement
Analysis across multiple mouse and human cell types identified two categories of chromatin movement. In the first, chromatin motion is constrained to about 200 nanometers over short to intermediate timescales (up to 200 seconds). This subdiffusive movement means loci are mostly in contact with neighboring genomic regions, facilitating rapid interaction with regulatory elements within roughly 100,000 base pairs—a timescale consistent with normal transcription.
The second category features more extensive chromatin movement over longer timescales (minutes to hours) and was observed in some but not all cell types. The reasons for this variability remain unclear.
Implications for gene regulation and modeling
The findings suggest a “region of influence” around each genomic locus, about 200 nanometers in size, within which loci can effectively interact. This has implications for understanding how genes find regulatory sequences and how broken DNA ends stay proximal for repair. The strength of the subdiffusive pull observed could not be fully explained by existing polymer models like the Rouse and fractal globule models, indicating a need to integrate nuclear environment interactions in future models.
Why it matters
Understanding chromatin dynamics at these detailed timescales improves the fundamental knowledge of gene regulation mechanisms and DNA repair. It also clarifies how genes and regulatory elements physically locate and interact with each other inside the crowded nuclear space, a process essential for normal cell function and response to DNA damage. The discovery that chromatin dynamics vary significantly between cell types could influence future research on cell-specific gene regulation and genome organization.
Background
Gene expression is regulated by interactions between genes and remote DNA regulatory elements positioned along the chromosome. Chromatin movement facilitates these contacts within the cell nucleus. Conventional imaging techniques have previously limited dynamic range measurements, hampering understanding of chromatin’s physical behavior. MINFLUX microscopy’s ultra-high resolution with long-term tracking capability enabled this breakthrough investigation of chromatin motion at the nanoscale and over extensive timescales.
Sources
This article is based on reporting and publicly available information from the following source:
Read more Science Discoveries stories on Goka World News.